For half a century, the notion of an artificial pancreas has tantalised researchers seeking to transform the management of people with diabetes. From scepticism to collaboration, from regulatory hurdles to clinical triumphs, the journey mirrors the resilient quest against diabetes. Unlike early backpack-sized prototypes, the new closed-loop system fuses a continuous glucose monitor with an insulin pump and a smart algorithm. Professor Roman Hovorka, the 2023 EASD–Novo Nordisk Foundation Diabetes Prize for Excellence laureate, has spearheaded the development of this technology, marking a paradigm shift in diabetes care.
For half a century, the idea of replacing a malfunctioning pancreas with an artificial one has persisted. This concept involves creating a device capable of replicating its vital function, insulin secretion, to effectively manage the blood glucose levels of individuals with diabetes, offering a potential life-changing medical solution. But developing this has been far from trivial.
“Insulin is a powerful drug. It is necessary for life. However, excessive insulin can lead to a coma or even be fatal. Therefore, we needed to ensure that the system was safe enough to progress to the next stage, where you can now take it home and incorporate it into daily life. Designing the system for children was even more challenging because food, and especially carbohydrates, affect the body more rapidly, and activity and eating patterns are less predictable,” explains Roman Hovorka, a Professor at the Wellcome-MRC Institute of Metabolic Science, Metabolic Research Laboratories at the University of Cambridge, United Kingdom.
Simple in principle
Roman Hovorka’s journey into the development of closed-loop systems was marked by a diverse background and a series of progressive steps. Beginning in the early 1980s, his career trajectory initially led him into the field of mathematical informatics, which he studied at Charles University in Prague, at that time in Czechoslovakia. His early exposure to the University Hospital in Prague in 1984, as the sole non-medical staff member, ignited his curiosity about the medical world, particularly diabetes.
This interdisciplinary background and curiosity led him to delve into the complex world of diabetes management and the development of closed-loop systems, bringing us closer to the realization of an artificial pancreas that can improve the lives of individuals with diabetes.
"I was part of a group that managed diabetes, and I was a person who was quite unique within the group. While I communicated using my mathematical background, they used the medical language. We had to find a way to communicate, but they could see the potential in what I could offer them at that time, in the mid-1980s. I could also see that I was able to help them solve problems during that period. This helped us move towards the concept of closed-loop systems."
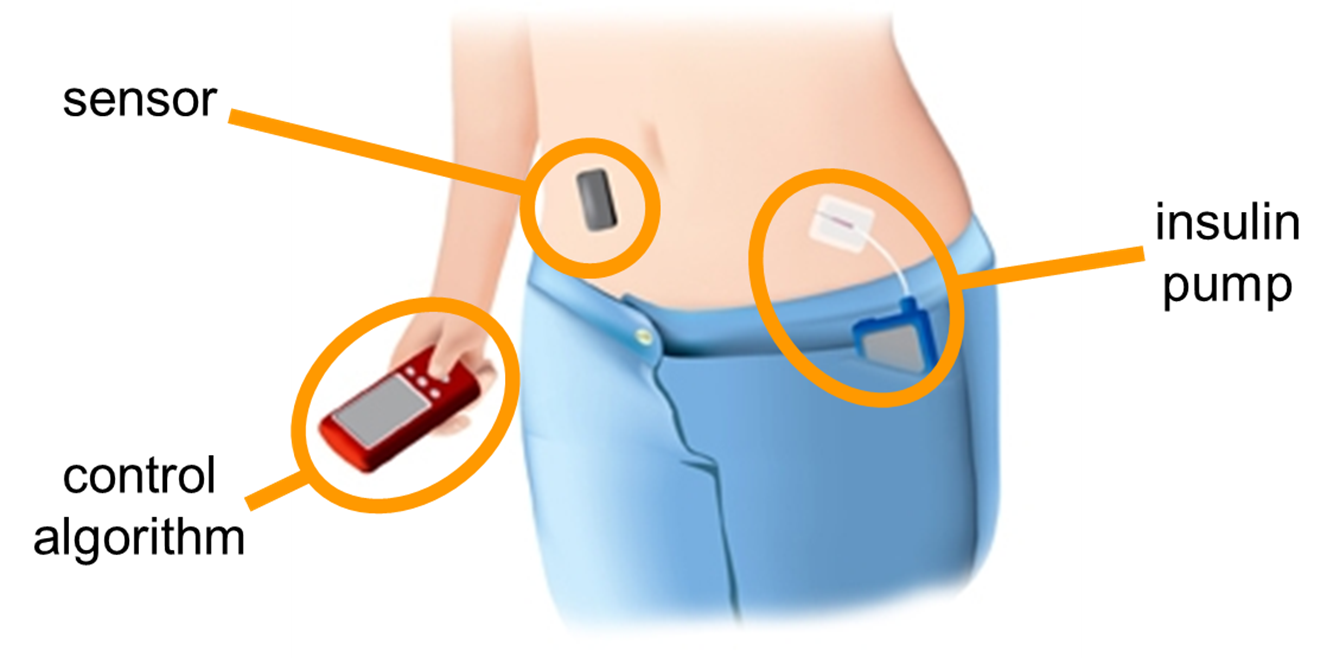
The idea of closed-loop systems is simple in principle.
"A continuous glucose monitor is a device that is placed on the skin, either on the arm or the abdomen. Through a little probe, it measures glucose levels every 1–5 minutes. These data are then transmitted to the computer algorithm, which processes the glucose values and instructs the insulin pump, which administers the required amount of insulin."
Roman Hovorka's interest led him to pursue a Ph.D. focused on creating a decision support system for individuals with type 1 diabetes. His career path took a pivotal turn when he secured a Wellcome Trust Visiting Fellowship, which facilitated his move to City University, London, in 1989. During that time, he continued to deepen his knowledge in pharmacokinetics, pharmacodynamics, physiology, clinical trials, and regulatory issues.
"I became involved in several European projects centered around diabetes management. Recognizing the potential for applying mathematical approaches to improve diabetes care, I became increasingly committed to advancing the field. And then, in the late 1990s, I got a call from a company called Disetronic, a Swiss-based manufacturer of insulin pumps, that was working on developing a closed-loop system."
Finding the way
However, the journey was not without challenges. In 2003, Roman Hovorka approached the Juvenile Diabetes Research Foundation (JDRF) with his vision for closed-loop systems despite their dedicated and longstanding focus on finding a biological cure.
"Their efforts at the time were not focused on devices but on cures such as immunotherapy or cell-based therapies. So at that time, I was forced to try to secure funding from other sources, which proved to be very difficult."
Up until then, the story of the closed-loop system had also been a story of broken promises.
"People kept estimating the solution would come in the next five to 10 years. And it never happened. And people were losing faith in that application, and trust was also lost."
"In 2006, the JDRF radically changed its position recognizing that a cure would not be immediately forthcoming, and if we have this number of people with type 1 diabetes, we need to help them. So even though the JDRF still maintains finding a cure is the ultimate goal, they appreciated the paramount importance of helping individuals who are already living with this condition and the potential of the artificial pancreas."
You cannot do everything on your own
The JDRF turned to their members, and the response was overwhelming. A substantial number of members wanted the JDRF to pursue solutions for those who already have type 1 diabetes. This prompted the establishment of the key JDRF Artificial Pancreas Consortium, which proved to be the turning point for the whole field.
"A group of leaders in the field was assembled, and substantial financial and support resources were allocated, including engagement with regulatory bodies. We found ourselves privileged to become part of the Consortium and to embark on the journey from that point forward."
The new Consortium brought about an important change.
"In the early days, people tried to do everything, from the pump to the algorithm and the sensor system. I think what people also realized at the time was that you need to collaborate. You can't do everything on your own. It's usual to have a sensor from one company and a pump from somewhere else. But the ability to put it all together was something that could accelerate progress."
In 2006, it was realized that it was more important to leave industry to handle the individual technological challenges and instead focus the new collaboration on the big gap, which was the algorithm.
Feedback and Motivation
Meanwhile, Roman Hovorka had moved from City University, London, to the University of Cambridge, where he set up his research group.
When the efforts to construct the closed-loop system had started, several people said that the researchers could never mimic the physiology of the pancreas – of delivering insulin to the portal vein and sensing glucose in the blood – and instead insulin should be administered, and glucose levels measured in subcutaneous tissues.
"There were people aiming for perfection. Initially, people actually tried to do an implantable system, where you implant the insulin pump and the sensor, but that was too difficult. This pursuit of perfection hindered progress. When I saw how people's diabetes was being managed, while it may not be ideal, it would be better to use an imperfect closed-loop system compared with what was available."
Roman Hovorka and colleagues talked to many people in the early stages of their research, including parents. They asked them whether they would trust a closed-loop system that injects insulin automatically for their children.
“We told them that, in principle, this is potentially dangerous because it can be life-threatening if the system fails. So we had this focus group with mothers and children, and many simply said, ‘We need anything. We're just tired. The burden of disease is too much on the family. We just need our lives back.’”
The feedback became the motivation and driver for the hard work to come. And probably in the same way that many people perceive mathematicians as highly theoretical individuals, Roman Hovorka approaches challenges differently.
“I have always been a problem-solving person who tries to see the problem and break it down into smaller problems. So it was a sort of stepwise process that benefited from lots of things I had learned in my early career. And, again, the motivation came from seeing a problem, being able to solve it, but also getting the feedback from people when they came back to us to say that what we are offering was life-changing.”

No two nights the same
The research, which started at the University of Cambridge in 2006, was divided into two main parts. One included safety studies carried out in a controlled environment in the clinical research facility of Cambridge Addenbrooke’s Hospital. People were closely monitored while the researchers tested the early version of the algorithm, which controls the insulin infusion.
“We were testing when study participants were running on a treadmill. We were testing big meals and small meals. We really pushed the system into the corners, which are difficult to manage. We were gaining confidence that the system could cope. We even had one study where adults drank three quarters of a bottle of wine because alcohol strongly affects glucose control, so we wanted to make sure that it worked even then.”
The many tests were also necessary because insulin is such a potent molecule, so they wanted to be sure that the system was safe enough to move to the next step, where they could confidently say that it was safe to use at home, on vacation, at school, skiing, or traveling.
“My main role was developing the algorithm, the computer program. And what it aims to do is basically describe how insulin affects glucose and, of course, other aspects like how meals affect glucose. And we aimed to create a model so we can better forecast glucose levels. We looked at the data coming from the individual patient to see which model would fit better to this person. And then we used all these models to optimize and time the insulin infusion.”
The second stage of the development started with shorter home studies: first three weeks, then three months, and eventually years, in which Roman Hovorka and his team tested various populations.
"We studied the closed-loop system in pregnancy; we tested it on very young children. And in those longer studies, we learned that every day and every night is even more different than we thought it would be. Therefore, we had to make the system more adaptive and more responsive. Because there’s a week and a weekend. There’s school and there’s a holiday. No two nights are the same.
Adapt 24 hours a day
The research showed that models could even change over time – and sometimes quite rapidly, so Roman Hovorka and his team had to figure out ways to jump from one model to another very quickly. The extensive tests taught them which parameters they had to change and how these parameters change.
“We were helped tremendously by also doing computer simulations. The keyword turned out to be adaptivity. So, we have developed a system that can adapt to people throughout the 24-hour cycle. The system continuously adapts; it never stands still.
Overnight glucose control is one of the major benefits of the artificial pancreas, especially for parents, who normally get up two to three times every night just to check that their child’s glucose levels do not get too high or too low.
"Every day and every night is, in principle, different. So what is good on one night is not good another night. And the closed-loop system, where the system is monitoring what’s happening regardless of the events of the day, can run on its own. This is where the closed loop kicks in: it will give the right amount of insulin every night, around the meals, throughout the 24-hour cycle. It is really the way forward before that elusive cure is found.”
The greatest life improvement with the closed-loop system is overnight.
“With a closed loop, your blood glucose is better controlled with fewer long-term side-effects, but it is the psychosocial benefits that are also important to families, both for the children and the parents. If you have better control, you have better sleep, mood, and concentration, and you feel safer – both as a child with diabetes and as a worried parent.”
Decided to set up his own company
With the development of a well-functioning closed-loop system, there would be no more guessing about how much insulin to give. The researchers did lots of studies, published widely, and carried out randomized controlled trials, which clearly showed the benefits. But despite the unanimous positive feedback from the users, one important hurdle was left: the regulatory approval of closed-loop systems to manage type 1 diabetes.
So what happened quite often in our clinical trials was, despite having bulky devices and not ideal usability for these devices, people were still willing to use them. And we did actually have some problems getting the devices returned because the participants had felt the positive change in their quality of life. But we couldn’t do it at the time because these devices were not regulatory approved and were still only usable under the Clinical Trials Directive.”
Roman Hovorka presented his research to the United States Food and Drug Administration, FDA, and United States National Institutes of Health, NIH, and to the European Commission’s advisory body on medical device regulation. His research was also presented on numerous occasions at leading international and national diabetes conferences including the American Diabetes Association, the EASD, Advanced Technologies & Treatments for Diabetes, and Diabetes UK.
“So we were able to work with the healthcare professionals and show them the system: this is what it can do. We were also preparing regulatory bodies: this is what’s likely to come in your direction.”
The initial intention from the researchers was to license the developed closed-loop system to established companies to take it to the market. There were positive discussions, but these failed to materialize. In 2018, Roman Hovorka and colleagues decided the only option was to set up a company, CamDiab Ltd.
“We started commercializing what we had done; it was a pretty big challenge at the time. But right now, we are in 15 European countries and Australia. We have over 16,000 users, and we are growing by about 1,300 users per month.”
Life-changing
Today, there are several commercial closed-loop systems, so the 60-year-old dream of creating an artificial pancreas is no longer just a dream, and Roman Hovorka is getting amazing feedback from people on their improved quality of life.
"Some people are saying that being on a closed loop is like taking a bit of a diabetes holiday. They can step back and not think about it."
For some people, it is a life-changing experience. So the differences are huge, especially for those people who are conscious about the parents who really care about their children and feel burdened by diabetes. We can take part of this burden from them, so they can spend more positive time on family life.
The journey has been long in getting to where the field is today. But Roman Hovorka feels privileged to be part of it.
“When I started working in this field, it was not something I could have imagined. I came with my mathematical informatics background. What was really crucial is that at my first job in the university hospital, I was dealing with people with real problems.”
Roman Hovorka has been along for the ride from the early safety studies, and later clinical studies, all the way to commercialization.
“We were able to go through the whole journey. I could see the world that was developed on paper, then on a computer, going all the way to reaching tens of thousands of people in real life. The motivation came from seeing a problem, being able to solve it, but also getting feedback from people who come back to you and say that this is life-changing.”
